A Helpful, Hopeful, and Evidence Based Guide
Age related macular degeneration, or AMD, affects millions of people. Many readers share the same question: Can stem cell AMD treatments really restore vision or stop the disease from getting worse?
There is a lot of hope around stem cell therapy for macular degeneration. But there is also a lot of confusion. Some people hear stories about miracle cures, while others see warnings about unsafe clinics. This guide brings together the latest information from trusted sources like NEI, NIH, ISSCR, and major research centers so you can feel confident and well informed.
The goal of this article is simple. Help you understand what is real, what is possible, and where stem cell therapy for macular degeneration is heading next.
First and foremost, do your due diligence, don’t rush to do a treatment that hasn’t been tested or at clinics that don’t specialize in AMD treatments.
What Stem Cells AMD Treatment Is Trying to Fix
Macular degeneration affects the center of the retina called the macula. This area helps you read, recognize faces, and see fine detail. In both dry and wet AMD, the cells in the macula become damaged. When these cells die, vision becomes blurry or distorted.
Stem cell therapy for macular degeneration aims to replace or protect these damaged retinal cells. Instead of trying to make the eye work harder, the goal is to provide new healthy cells that support the retina.
Researchers hope this approach could slow the disease, stop further damage, or in some cases restore a small amount of lost vision.
Current State of Stem Cell Research for AMD
What Scientists Are Studying
Most research focuses on replacing damaged retinal pigment epithelium cells, often called RPE cells. These cells keep photoreceptors healthy. When RPE cells fail, vision declines.
Types of Clinical Trials Happening Now
Researchers around the world are studying stem cells for macular degeneration in several ways:
- RPE replacement using human embryonic stem cells
• RPE replacement using induced pluripotent stem cells
• Adult stem cell therapy for macular degeneration to protect remaining cells
• Stem cell derived patches implanted under the retina
• Gene edited stem cells to make them safer and more stable
Where Research Stands in 2025
Most trials are in Phase 1 or Phase 2. This means researchers are learning about safety and early effectiveness. Phase 3 trials, which lead to FDA approval, are still limited.
FDA Approval Status
As of 2025, no stem cell therapy for macular degeneration is FDA approved. All available treatments are still experimental and only available through regulated clinical trials.
A Quick Snapshot of Progress
- Early trials show that transplanted RPE cells can survive in the eye
• Some patients regain small amounts of vision
• Serious complications are rare when done under strict research protocols
• Larger studies are underway in the United States, Japan, South Korea, and the United Kingdom
• FDA approval is unlikely before 2028 to 2030 based on normal research timelines
Simple Table of Current Major Trials
|
Research Group |
Type of Stem Cell |
Trial Phase |
Key Findings |
|
NEI/NIH |
Induced pluripotent stem cell derived RPE |
Phase 1 |
Safe so far, stable cell survival |
|
Japan RIKEN Team |
iPSC RPE sheet |
Phase 1 |
Vision stable or slightly improved |
|
Astellas Pharma |
Embryonic stem cell RPE |
Phase 1/2 |
Some vision improvement reported |
|
London Project to Cure Blindness |
Stem cell RPE patch |
Phase 1/2 |
Patients regained reading ability |
|
US Academic Centers |
Adult stem cell intravitreal injections |
Early stage |
Focus on slowing progression |
A common question I get is “Does the stem cell treatment last?” The answer is yes, the most current studies are showing up to 3 years of lasting results. It is still new so longer test periods are ongoing.
Types of Stem Cells Being Studied
Researchers are studying several types of stem cells for macular degeneration.
1. Embryonic Stem Cells
- Can become any cell type
• Often used to create RPE cells
• Strong potential but raises ethical questions
• Requires strict regulation
2. Induced Pluripotent Stem Cells
These are adult cells turned back into stem cells.
• No ethical controversy
• Can be customized using a patient’s own cells
• Lower chance of immune rejection
• Currently one of the most promising options
3. Adult Stem Cells
These come from bone marrow, fat tissue, or blood.
• Safer and easier to obtain
• Less powerful than embryonic stem cells
• May help protect cells rather than replace them
What Doctors Most Often Use in AMD Trials
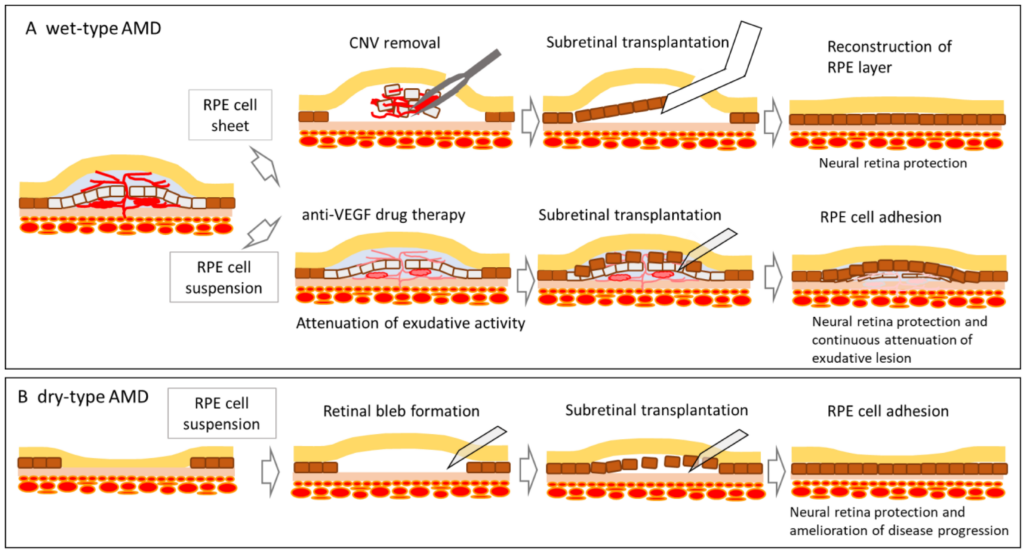
RPE replacement patches made from induced pluripotent stem cells or embryonic stem cells. These cells are grown in a lab and placed under the retina using delicate surgery.
How Stem Cells Are Delivered
- Injection into the eye (less common today)
• RPE cell sheets or patches placed under the retina
• Tiny surgical tools that slide the patch into position
• Protective medications to prevent rejection
Imagine replacing worn out tiles in a small area of the floor. You carefully slide in a new tile so it fits perfectly. That is similar to how stem cell patches are placed under the retina.
Safety and Risks of Stem Cell Therapy for Macular Degeneration
What Clinical Trials Show
In reputable trials, complications have been uncommon. Researchers carefully check for:
- Inflammation
• Infection
• Scar tissue formation
• Immune rejection
• Abnormal cell growth
One of the biggest risks is the possibility of tumors if cells are not prepared correctly, which is why regulated trials follow strict guidelines.
Why Unregulated Stem Cell Clinics Are Dangerous
Some clinics in other countries promise instant results. These clinics often charge large amounts for treatments that are not proven or safe. People have lost vision from unregulated injections. There is a risk for stems to grow out of control causing tumors or cancer.
If anyone asks where can I get stem cell treatment for macular degeneration, the safest answer is always a regulated clinical trial listed with FDA or NIH.
Risk Comparison Chart
|
Treatment Type |
Main Risks |
Safety Notes |
|
Clinical trial stem cell therapy |
Mild inflammation, rare surgical risks |
Strong safety monitoring |
|
Standard AMD injections |
Bleeding, infection, pressure rise |
Very common, well understood |
|
Unregulated stem cell clinics |
Vision loss, retinal damage, infection |
Not recommended |
Who Might Qualify for Stem Cell Trials
Typical Inclusion Criteria
- Dry AMD with geographic atrophy
• Early to moderate wet AMD in some studies
• Vision not yet completely lost
• Overall good health
• Ability to attend many follow up visits
Typical Exclusion Criteria
- Active infection
• Other major eye diseases
• Severe bleeding disorders
• Recent major surgeries
• Poor general health
Realistic Expectations
Stem cell therapy for macular degeneration does not currently cure AMD. It may:
- Slow progression
• Maintain current vision
• Provide small improvements
• Help certain cells stay healthy longer
A simple example:
A patient in a UK trial reported going from only seeing shapes to being able to read small print again with the help of a handheld magnifier. The improvement was small but meaningful.
Dry AMD and Stem Cells
Dry AMD is the main target of research. This is because RPE cells slowly break down over time. Replacing these cells could help slow or stop the progression of geographic atrophy. A common question I get is “ Can stem cells reverse macular degeneration?” The answer is that current clinical trials are showing positive outcomes in slowing the progression and in some cases reversing the damage in AMD
What Trials Show
- RPE patches stay in place
• Atrophy may slow down
• Some patients see letters more clearly
• Most improvements are modest
Wet AMD and Stem Cells
Wet AMD involves leaking blood vessels. Stem cell therapy is currently used only after anti VEGF treatments have controlled the leakage.
What Researchers Hope
- Healthy RPE cells may reduce the need for injections
• Stem cell patches could stabilize the retina
• Trials are still very early
Costs and Accessibility of Stem Cell Therapy
-
How Much Does It Cost
If you are part of a clinical trial, treatment is usually free. Some travel or lodging costs may not be covered.
The cost of stem cell therapy for macular degeneration outside trials can be extremely high, often more than 10,000 dollars for unproven treatments. These are not recommended.
-
Insurance Coverage
Insurance does not currently cover stem cell therapy for macular degeneration because it is still experimental.
-
How to Find a Legitimate Trial
Use official sources like:
- clinicaltrials.gov
• NIH websites
• Academic eye centers
• ISSCR research listings
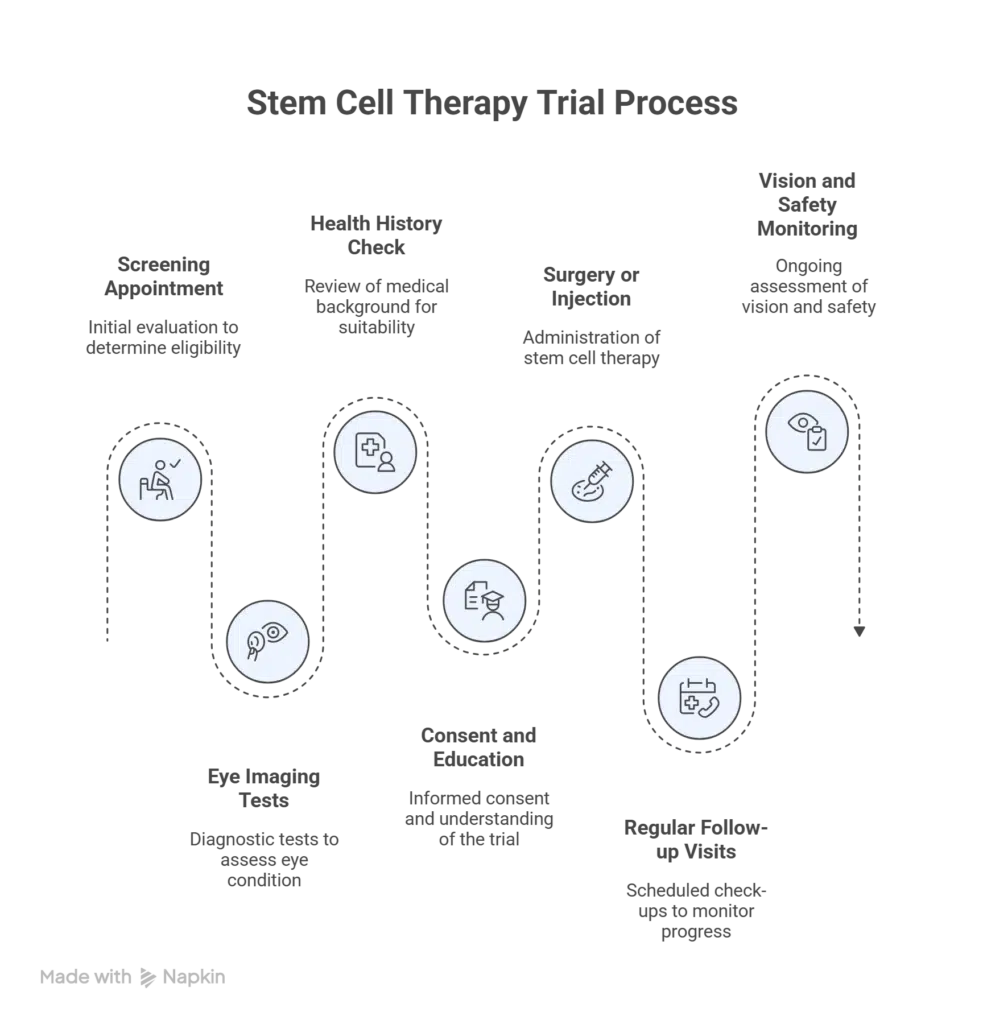
Patient Journey Map
A simple step by step overview of what happens in a trial.
- Screening appointment
- Eye imaging tests
- Health history check
- Consent and education
- Surgery or injection
- Regular follow up visits
- Vision and safety monitoring
Many patients report that the process feels comfortable and well supported, especially at major research centers.
Research Timeline
- 2006: iPSC discovery
• 2010: First lab grown RPE cells
• 2015: First human iPSC retinal transplant in Japan
• 2018: London Project to Cure Blindness showed reading improvements
• 2020 2024: Multiple Phase 1 trials completed safely
• 2025: New Phase 2 trials underway across several countries
• 2028 2030: Possible FDA review if trials succeed
Helpful Tools for People Living With AMD
While waiting for new treatments to become available, many people use vision tools at home.
- Strong handheld magnifiers
• Portable electronic magnifiers
• Full size desktop CCTVs
• Large print materials
• High contrast lighting
If you are exploring tools that make daily life easier, you can find friendly guides at Lowvisionaids.org, including options like handheld magnifiers and other low vision products.
A simple example from a customer story:
A reader said, My new portable magnifier helped me keep reading recipes until my trial visit. It made a big difference in my confidence.
Another patient said: “Having bioptics on my glasses made it so I could keep driving while I’m in the clinical trial for stem cells, they’ve been a life saver!” Maria R.
Frequently Asked Questions
-
Is there a cure for macular degeneration stem cells can provide
Not yet. Current treatments focus on slowing the disease and possibly improving small amounts of vision.
-
Are stem cells safe for macular degeneration
In regulated trials, safety looks promising. In unregulated clinics, the risks are high.
-
Where can I get stem cell treatment for macular degeneration
Only through official clinical trials. Avoid private clinics that claim immediate results.
-
How long until stem cell therapy is available for everyone
Most experts estimate several more years. Large Phase 3 trials must be completed first.
-
Can stem cells help both dry and wet AMD
Yes, but research is most advanced for dry AMD.
- Are there many risks with stem cell treatment for AMD?
The answer is that there is risk with any surgical procedure. There have been early study patients that lost vision due to their procedures. Its very important to participate in strict, well funded and controlled trials.
Final Thoughts
Stem cells offer real hope, but they are not yet a cure. The research is moving forward, and the results so far are encouraging. If you want to understand your options, your eye doctor can help you find legitimate trials and supportive tools.
For readers living with AMD right now, practical tools like magnifiers and CCTVs are still the most effective way to make daily life easier. LowVisionAids.org offers simple guides and supportive resources for people with low vision, caregivers, and families.
Don’t hesitate to reach out to us for help, we can help you choose the right device to keep you doing the things you love.
You are not alone, and new possibilities are coming.