Many people worldwide suffer from the degenerative retinal disease, which may result in blindness. The main cause of blindness in Caucasians older than 40 in the USA is age-related macular degeneration. The main cause of vision loss in those between the ages of 20 and 74 is diabetic retinopathy. One of the most frequent causes of hereditary retinal diseases that result in blindness is retinal pigmentosa, which affects 1 in 3000-7000 individuals.
Anti-VEGF or Anti-vascular endothelial growth factor intravitreal injections are now the only FDA (Food and Drug Administration) authorized therapy for diabetic retinopathy complications and neovascular AMD. Similar to how anti-VEGFs and laser photocoagulation are used to treat diabetic retinopathy. Although they are successful in addressing the side effects brought on by these disorders, they have little ability to change the course. Retinitis pigmentosa (RP) therapy up until recently consisted of steps to stop more damage or halt the illness. However, the RPE65-targeting gene therapy Luxturna (voretigene neparvovec-rzyl) has acquired FDA clearance.
These degenerative retinal disorders may be treated using gene and stem cell therapy. An effort has been made to create innovative treatments that include the substitution of mutant genes, immunomodulation, extended administration of neurotrophic factors and/or medication delivery, regeneration of atrophic or injured retinal tissue, and immunomodulation by viral vector delivery. The goal of this study is to describe the retinal problems and illnesses that affect patient groups most often and to examine some of the cutting-edge therapeutic options presently being researched, such as the use of stem cells and gene therapy methods.
Visual morbidity from degenerative retinal degeneration is considerable everywhere. In the industrialized world, diabetic retinopathy is one of the main causes of blindness. Stem cell and gene therapies may be able to cure the consequences of this and other degenerative retinal disorders, even while present treatments only halt the disease’s course. The use of various stem cell types for the regeneration of atrophic or damaged retinal tissue, the lengthy administration of neurotrophic factors and/or drug delivery, immunomodulation as well as the replacement of mutant genes, and immunomodulation via viral vector delivery are some of the novel therapies currently under investigation.
BetaStem therapeutics diabetic retinopathy study
Dr. Stephen Bartelmez is the CEO of the stem cell treatment organization BetaStem Therapeutics. Their goal is to cure diabetic retinopathy by repairing the retinal blood vessel damage brought on by diabetes utilizing the body’s own stem cells.
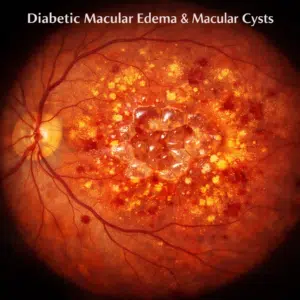
The retina, which is found in the rear of the eye, contains the macula. Despite being quite little (approximately 5 mm in diameter), it is responsible for the majority of human vision, including color perception and fine detail. Diabetic macular edema is a swelling of the retina brought on by fluid leaking from diabetic-related blood vessel damage in the macula. Additionally, diabetic retinopathy (DR), or damage to the blood vessels in the rear of the eyes, may develop over time as a consequence of macular ischemia (inadequate blood flow). There is presently no effective therapy to restore blood vessels and cells that have been harmed by DR, one of the primary causes of adult blindness.
Loss of eyesight ultimately results from retinal injury.
Cells originating from circulating CD34+ stem cells regularly rebuild blood arteries in healthy individuals. These stem cells have the protein CD34+ on their surface and are produced in the bone marrow before being released into circulation. In addition to producing endothelial cells, the basic units of all blood arteries, they divide and develop into all nine other kinds of blood cells. Endothelial cells and stem cells go to blood vessel lesions to aid in healing. However, as diabetes progresses, the patient’s CD34+ stem cells lose their ability to function, making it impossible for them to produce endothelial cells or control the healing of damaged blood vessels in the retina or the rest of the body.
Transforming growth factor beta 1 and stem cells
Haematopoiesis, the process by which all blood cells are produced, begins with haematopoietic stem cells. A healthy adult produces between 50 and 70 billion new blood cells a day, of nine distinct kinds, in the bone marrow, where this occurs. A complicated chain of actions including several signals from the surrounding tissue as well as inside the stem cells themselves is necessary to produce all these cells.
Retinopathy may lead to blindness or vision loss.
A tiny protein called Transforming Growth Factor beta 1 (TGF-1) is released by several bodily cells. Dr. Bartelmez and colleagues discovered that TGF-1, which is categorized as a cytokine (a kind of tiny protein crucial in cell signaling in blood and immune cells), is a key regulator of haematopoiesis. Cell divisions at important cellular checkpoints are finely inhibited by TGF-1. Their research has made it clearer how TGF-1 affects stem cells in the human bone marrow and blood.
TGF-1 regulates all phases of hemopoiesis.
The researchers were successful in separating CD34+ stem cells from other blood cells present in mouse and human blood samples and growing them in the lab. Next, the researchers were able to temporarily suppress TGF-1 in the stem cells. A PMO is a tiny DNA mimic that momentarily prevents stem cells from producing a certain protein. The researchers investigated many methods for introducing PMO into cells, including syringe loading, microinjection, and pairing PMO with cationic lipids, which are lipids with an electrical charge. However, it was shown that unaided access was the most effective way to absorb TGF-1-PMO into stem cells. Thus, TGF-1-PMO was picked up by endosomes on the surface of CD43+ stem cells following overnight incubation with the PMOs at 37 degrees Celsius. It was discovered that both the incubation temperature and PMO concentration were crucial.
Stem cells that are actively proliferating
In fact, this method momentarily suppresses TGF-1 in CD34+ stem cells. It functions as a switch to first block TGF-1, and then the stem cell responds to this blocking by upregulating the TGF-1/TGF-mRNA gene. The PMO is then effluxed from the stem cell, resulting in an increase in TGF-1 protein synthesis.
Numerous uses for the protein have been discovered. Cells may be encouraged to divide or prevented from doing so; they can be encouraged to self-destruct or differentiate (become a new kind of cell), or these chemical processes can be stopped from happening. Transient TGF-1 suppression establishes a balance between CD34+ cell growth, maturation, division, and times of cell inactivity, according to one important discovery.
Different types of stem cells
While there is some disagreement in the definitions offered, stem cells are often understood to be groups of cells that are both self-renewing and capable of differentiating into a variety of cell types. Depending on the circumstance, these cells are referred to as multipotent or pluripotent. Thought to be incapable of regeneration, the mature retina of mammals is now known to contain a population of retinal stem cells that are confined to the pigmented ciliary margin and capable of differentiating into a variety of retinal cells, including rod photoreceptors, bipolar cells, and Müller cells. Since then, these cells have been labeled as pigmented ciliary epithelial cells or late-stage neural progenitors. As a specialized branch of the nervous system, the retina, and neural progenitor/stem cells are crucial to the development of the retina.
Many people have speculated that the injured or degenerating retina might have regenerative potential via the transplantation or activation of stem cells since the multipotent property of the progenitor cells is vital to the normal development of the structures in the eye and retina. The first efforts at introducing neural and retinal stem cells into the deteriorating retina failed because these cells could not integrate into the retina or restore eyesight. However, MacLaren et al. observed that rod photoreceptor precursors produced from the post-natal day 1 (P1) retina of mice may be incorporated into the outer nuclear layer of the mammalian retina (ONL). In mice models of degenerative retinal degeneration, they saw that these cells developed, created functional connections, and enhanced vision. The inference made was that the integration of the stem cells into the retina depends critically on the ontogenetic stage of the precursor cells, in this instance as indicated by the expression of neural retina leucine zipper (Nrl).
Others have noticed that the transplantation of neural progenitors produced from embryonic stem cells improves the maintenance of visual function and the survival of host retinal cells such as photoreceptors in the mind mice model of neuronal ceroid lipofuscinoses. Given that treatment of IGF-1 has been shown to have effects comparable to those described above, it has been hypothesized that this improved host cell survival may be caused by the production of growth factors.
Stem Cell Treatments focused on Diabetic Retinopathy
Although the current clinical categorization is mostly based on microvascular lesions, it is well known that diabetes damages the retina’s vasculature, neurons, and glia. Retinal neurodegeneration is now well acknowledged to have a substantial role in DR.
These include reactive gliosis, decreased neuronal function, glutamate excitotoxicity, decreased levels of neurotrophic factors, and neural accelerated apoptosis, which mostly impacts retinal ganglion cells and amacrine cells, but photoreceptors may also be affected. In fact, all of the retina’s primary cell types have been proven to be changed, and endothelial cells, neurons, microglia, astrocytes, and Müller cells may interact to cause DR.
In addition, alterations in DR are believed to contribute to the disease in the choroid’s vasculature, which feeds the outer retina, photoreceptors, and retinal pigment epithelium (RPE). Additionally, there is proof that peripheral immune cells, such as neutrophils and monocytes, are activated in DR, increasing leukocyte-endothelial interaction and the risk of capillary occlusion and the release of pro-inflammatory cytokines, both of which contribute to increased vascular permeability.
The neurovascular unit’s dysfunction is a crucial aspect of DR, although, for patients who are still in the early stages of the illness, its effects on retinal function are mostly asymptomatic. Retinal ischemia and hypoxia are caused by the disease’s growing capillary non-perfusion, which in turn causes pro-angiogenic proteins like vascular endothelial growth factor (VEGF) to be upregulated and inflammatory pathways to be activated.
Some individuals eventually develop severe, sight-threatening phases of diabetic macular edema (DME) and proliferative DR as a result of retinal ischemia and high levels of VEGF, which causes disruption of the blood-retinal barrier, excessive vasopermeability, fluid buildup, and neovascularization (PDR). Greater and more targeted therapy approaches could result from a better knowledge of the underlying disease processes and the interaction between intrinsic vascular cells, circulating cells, and neural factors in the retina and choroid.
The majority of currently available treatments, including anti-VEGF injections, corticosteroids, vitreoretinal surgery, laser photocoagulation, and photocoagulation, target the disease’s latter stages when considerable damage has already been done.
Recent research also suggests that using anti-VEGF injections at an early stage, before DR problems manifest, may prevent the development of severe phases. The management of DR has been transformed by anti-VEGF medication, and many patients now benefit from it. However, not all patients benefit from anti-VEGF medication, and in certain cases, difficulties might arise as a consequence of it. The pricey therapy, which patients must endure repeatedly over an extended period of time, strains the capabilities of all healthcare systems.
There are currently very few treatment alternatives available to stop the development of DR from its early stages to its sight-threatening stage. Fenofibrate has the potential for this use and has a license in several nations to stop DR development. However, further proof is required.
Tight management of glycemia, dyslipidemia, and hypertension are additional ways to lower the chance of developing DR or stop its development. Numerous pre-clinical studies in diabetic mouse and rat models have demonstrated the potential of neuroprotective therapies for the treatment of DR, and the administration of neuroprotective agents such as insulin-like growth factor 1 (IGF-1). Brimonidine and somatostatin, two putative neuroprotective agents, seemed to slow the course of retinal functional abnormalities in patients with pre-existing neurodys function, even though a clinical study utilizing these medicines failed to find differences across groups or when compared with a placebo.
Reperfusion Of Retina’s with Diabetic retinopathy
A large number of researchers are now looking at the possibilities of regenerative medicines for the treatment of DR. Case reports of patients having spontaneous reperfusion of an ischemic (lack of oxygen) retina and recovery of visual acuity provide credence to the concept that retinal function may be recovered by regenerating retinal blood flow. There have also been reports of spontaneous reperfusion in diabetic ischemic retinas. Through recurrent fluorescein angiography, Muraoka et al. observed a comparatively high rate of revascularization of nonperfused regions (in 40 of 60 eyes with DR).
For the purpose of encouraging vascular repair in pathological circumstances and preserving vascular homeostasis, vessel resident endothelial progenitor cells are crucial. Progenitors may become active during vascular damage, aid in endothelial regeneration, and encourage perfusion restoration.
Mice used in lineage tracing studies have shown that vessel-resident endothelial progenitors contribute to the development of neovessels during wound healing. Diabetes mellitus is widely known to cause chronic wounds with poor wound healing, which indicates abnormalities in normal repair mechanisms and may potentially affect endogenous vascular repair systems. A unique treatment approach could be to improve or restore endogenous healing systems. On the other hand, stem cell treatment provides promise for restorative therapeutic strategies in DR. A variety of stem cells have been examined in pre-clinical models of DR (MSC). The choice of the most promising stem cell product is still difficult because to DR’s complexity. Furthermore, it is still challenging to identify the patients who are most in danger of losing their vision and those who may benefit from restorative treatment. Finally, further study is required to pinpoint the precise moment to start therapy for the greatest results.
A summary in Layman’s terms
Stem cell therapy for Diabetic retinopathy is being studied extensively and is showing great promise. There are several mechanisms that may be used. Early findings have shown that spontaneous reperfusion of the retina is possible with the recovery of some visual acuity. It is still early in the quest to find the right solution with safe repeatable outcomes that is also an efficient way to treat a patient.
FAQ’s
Can diabetic retinopathy be cured with stem cells?
Mesenchymal stem cell transplantation may postpone the onset and progression of diabetic retinopathy. Early retinal problems in diabetic mice may be treated with adipose-derived stem cells and released paracrine substances.
Are stem cells able to restore vision?
Without surgery, stem cell treatment may improve your eyesight and stop further loss. It is a fantastic alternative for those who are wary of having their eyes operated on due to potential hazards. In order to recover your eyesight and avert eventual blindness, stem cells can regenerate retinal pigment cells.
What treatment for diabetic retinopathy is the most efficient?
- Laser therapy (photocoagulation).
If laser therapy is administered before the retina has suffered significant damage, it often prevents visual loss.
- Scatter (pan-retinal) photocoagulation is a more intense laser treatment that may be used to treat severe proliferative retinopathy.
- Anti-VEGF injections
- Stem cell therapies show promise.