Macular degeneration is a disease that attacks the center of your retina known as the macula, leading to central vision deterioration while peripheral (side) vision remains clear. There are two forms of macular degeneration; dry and wet.
Early stage dry AMD patients can often slow the progression of their disease with vitamin supplements and diet rich in natural antioxidants found in fruits and vegetables such as lutein and zeaxanthin, like vitamins.
1. Vitamins and Minerals
Dry macular degeneration occurs when the macula, the part of your eye that allows you to see fine details such as straight lines and colors, becomes damaged.
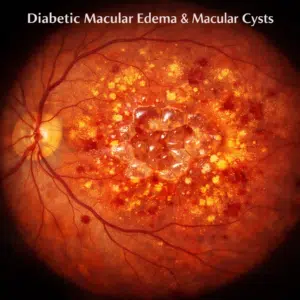
AMD can occur as the result of aging and the gradual thinning of retinal tissues, the layer at the back of your eye that transmits light signals to your brain. Over time, this can lead to blurry spots in central vision; usually beginning as tiny deposits called drusen that form underneath your retina – though usually these don’t interfere with vision loss directly; your doctor can use a special grid to help monitor any changes to your central vision.
When suffering from wet AMD, abnormal blood vessels grow beneath the retina and leak fluid or blood, leading to severe vision loss over time. While less common than dry macular degeneration, wet macular degeneration is more serious and less common overall; its cause could include genetic disorders or environmental influences.
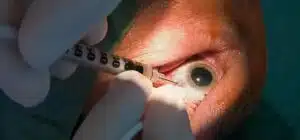
Anti-VEGF injections into the eye can prevent new blood vessels from growing and leakage by regularly administering this medicine for multiple years with your physician. These injections should be given on an annual basis.
Photodynamic therapy and thermal laser treatment may also help treat wet macular degeneration by decreasing leakage from new blood vessels and improving central vision.
Diets rich in leafy green vegetables, berries, tomatoes and other vibrant fruits and vegetables can help prevent or slow AMD. These foods contain antioxidant vitamins which protect eyes from oxidation and decrease visual acuity.
Your doctor can suggest specific vitamin and mineral supplements to treat dry eye macular degeneration, such as lutein, zeaxanthin, zinc and vitamin E – found in vegetable oils, wheat germ and almonds – that will help neutralize unstable molecules that break down healthy tissue, such as those found in vegetable oils or wheat germ. Omega-3 fatty acids found in fish such as salmon or tuna also reduce inflammation while increasing tear production.
2. Injections
Macular degeneration usually progresses slowly over time and allows patients to maintain central vision – this form is known as “dry macular degeneration”. Dry AMD is marked by the accumulation of deposits called “drusen” beneath the retina; they can be detected during dilated eye exams by looking for black lines arranged grid-like on white part of eye called an Amsler Grid. Ten to fifteen percent of patients may develop wet form macular degeneration where blood vessels form beneath retina and leak fluid directly onto it; rapid and severe loss of central vision ensues due to rapid blood vessel formation under retina, with abrupt and severe loss of central vision loss as a result.
Wet macular degeneration is characterized by abnormal blood vessels under the retina that leak fluid, killing macula cells and ultimately leading to blindness in the United States. Symptoms of wet macular degeneration are sudden and severe, including sudden blind spots or blurred areas near the center of your vision, distortion of straight objects such as telephone poles or venetian blinds appearing crooked, difficulty reading, driving or recognising faces, as well as difficulty with reading, driving and recognizing faces. Although wet macular degeneration is more serious than dry macular degeneration, most individuals who get it retain peripheral vision intact.
Up until recently, laser photocoagulation was the only treatment available for wet macular degeneration; using pain-free laser light to destroy leaky blood vessels which cause macular degeneration. Although effective at reducing vision loss in most cases, it could not restore lost central vision. Recently, injections of Visudyne were utilized as part of a technique known as photodynamic therapy (PDT). Once administered through injection into an arm vein, Visudyne could then be activated using non-thermal laser light on retinal blood vessels in order to provide photodynamic effects. Visudyne laser treatments close blood vessels and trigger chemical reactions that dissolve abnormal ones, leading to improved vision for about 15% of its treated patients while slowing progression of wet macular degeneration in others.
3. Laser Surgery
The macula is the area in the center of your retina that enables you to clearly perceive fine details when driving or reading, such as faces or small details. As we age, macular degeneration causes our maculas to thin and break down, leading to blurry or distorted central vision and potentially blurry or distorted side vision as well. While peripheral (peripheral) vision remains unaffected by macular degeneration symptoms may make it hard for you to see straight ahead when viewing faces or small details; blind spots may even appear depending on which type of macular degeneration you suffer from; depending on which type it may improve or worsen over time depending on its severity and type.
Dry macular degeneration, the most prevalent form of macular degeneration, occurs when parts of the macula become thinner and tiny yellow deposits known as drusen form under the retina. You may begin to notice straight objects appear crooked or blurry while colors appear less vibrant. Dry macular degeneration may progress to wet macular degeneration where abnormal blood vessels form beneath the retina that leak fluid that damages macula, though wet macular degeneration occurs more rarely but may quickly cause central vision loss.
There are various treatments for wet macular degeneration available today, including drugs that reduce the formation of new blood vessels while slowing their leakage of fluid from existing ones – known as anti-VEGF treatments and administered via eye injections. One such anti-VEGF therapy was FDA-approved with Visudyne’s photosensitizing drug activated via laser beam which destroys any new neovascularization; there may also be similar research underway with other drugs using similar principles.
Macular degeneration cannot be reversed completely, but you can slow the progression by eating a diet rich in dark green vegetables, fish and nuts and engaging in regular physical exercise. Routine eye exams with dilation as well as quitting smoking will also help. Individuals at higher risk for developing macular degeneration should also take vitamin supplements that include antioxidants and zinc for prevention.
4. Eye Drops
Macular degeneration affects the macula, the part of your retina responsible for clear central vision. AMD destroys this part, which makes reading or driving a car increasingly difficult. However, peripheral vision remains undisturbed; you will still see things like clock faces, your hands or faces of people nearby.
Macular degeneration usually progresses slowly and doesn’t result in complete blindness, with initial signs such as gradual loss of straight lines in one or both eyes, blurriness of central vision or distortions to geometric forms as indicators of macular degeneration. Your doctor can diagnose your condition using an Amsler grid chart; alternatively they may perform optical coherence tomography (OCT) testing or fluorescein angiography tests which involve injecting harmless orange-red dye into veins in your arm and following it through veins under retina where your doctor can observe blood vessels for signs of bleeding or leakage in order to detect and monitor them before further action can be taken against.
ARMD can take two forms. The vast majority is the dry form, which involves the gradual thinning and breakdown of macular tissue and accumulation of yellow deposits known as drusen under the retina. Meanwhile, 10% are wet forms which feature abnormal blood vessels growing beneath the retina that leak blood and fluid into the macula, leading to severe vision loss.
Many individuals with wet macular degeneration can successfully restore some vision after receiving treatment; however, central vision may never return fully. Common forms of treatment for wet macular degeneration include injections, photodynamic therapy and surgery.
Regular visits to an eye care professional are essential in the battle against macular degeneration, both prevention and treatment. At these visits, your doctor will use dilation eye drops on you in order to assess changes related to macular degeneration in the back of your eyes. However, please remember that these eye drops should never be used without follow instructions in order to avoid infection.