Glaucoma medicine: The possibilities for treating glaucoma are many. These consist of surgery, laser treatments, and eye drops. All of them aim to lower eye pressure and safeguard the optic nerve in the process.
Currently, ocular drops are frequently the primary option for treating patients in the US. Many patients can safely manage their ocular pressure for years with a mix of medicines and laser therapy.
Glaucoma eye drops work to lower eye pressure by improving the drainage of the eye’s fluid and/or reducing the amount of fluid produced by the eye.
The active component of glaucoma medications is used to categorize them. These include rho kinase inhibitors, alpha agonists, beta blockers, prostaglandin analogs, and carbonic anhydrase inhibitors. For individuals who need more than one type of therapy, combination medications are also an option. The cholinergic agonists, an earlier family of drugs (including pilocarpine), are no longer widely utilized because of their negative side effects.
Glaucoma Eye Drops Types
Xalatan® (latanoprost), Lumigan® (bimatoprost), Travatan Z® (travoprost), ZioptanTM (tafluprost), and VyzultaTM (latanoprostene bunod) are examples of prostaglandin analogs that function by enhancing the outflow of fluid from the eye. They are connected with changes to the eye itself, such as iris color changes and eyelash development, but they have little systemic adverse effects. One of these treatments could be more efficient and have fewer adverse effects depending on the patient. There are now generic versions of latanoprost and a few variations of bimatoprost. Tafluprost is a prostaglandin analog without preservatives.
The second-most popular family of medications, beta blockers, including timolol, act by reducing fluid output. They can be purchased in generic form, which may result in a lower price. There is also a version of timolol without preservatives. By shutting the eyelids after applying a beta blocker or by adopting a method termed punctual occlusion, which stops the medication from reaching the tear drainage duct and systemic circulation, systemic adverse effects of beta blockers can be reduced.
Alpha agonists [Alphagan®P (brimonidine), Iopidine®] function to both promote drainage and reduce fluid production. The purite preservative in Alphagan P breaks down into components of natural tears and may be better tolerated by persons who are allergic to other eye drops’ preservatives. The generic version of Alphagan is offered.
Inhibitors of carbonic anhydrase (CAIs) lower eye pressure by reducing the flow of intraocular fluid. These are available as tablets [Diamox (acetazolamide) and Neptazane® (methazolamide)], as well as eye, drops [Trusopt® (dorzolamide), Azopt® (brinzolamide). All CAIs, with the exception of brinzolamide, are accessible in generic form.
The outflow of intraocular fluid is accelerated by rho khinase inhibitors like Rhopsera® (netarsudil). Since April 2018, a new class of glaucoma medications has been accessible.
Patients who require many types of medication may have an alternative to combined drugs. Utilizing a single eye drop vial as opposed to two reduces preservative exposure in addition to convenience. Depending on your insurance plan, there can also be a financial benefit. Timolol, a beta blocker, and dorzolamide, an inhibitor of carbonic anhydrase, make up the drug Cosopt®, which is also offered in generic and preservative-free forms (Cosopt® PF). Brimonidine, an alpha agonist, and beta blockers are combined in Combigan® (timolol). Brimonidine and brinzolamide make up the combination drug Simbrinza®, which does not contain beta blockers.
Medication side effects for glaucoma
ALPHA-2 ADRENERGIC AGONISTS
The formation of aqueous humor is decreased by alpha agonists, which lower IOP. A popular selective alpha-2 adrenergic agonist is brimonidine. Six to nine months after the start of the medication, brimonidine can result in acute follicular conjunctivitis in 5% to 9% of individuals. After 15 months, a delayed follicular response has been seen. Another alpha-2 adrenergic agonist, apraclonidine, have a greater incidence (30-48%).
Alpha-2 adrenergic agonists frequently cause dry mouth, which is a side effect that can be related to a systemic action. The lipophilic nature of this medication family and its capacity to pass the blood-brain barrier may make children and older people more susceptible to central nervous system side effects including lethargy, apnea, hypotension, and bradycardia.
Occasionally, brimonidine can produce granulomatous uveitis that goes away after the medication is stopped.
Using a conjunctival biopsy, this medication has also been linked to ocular lichen planus, which can be treated with topical cyclosporine.
BETA BLOCKERS
Topical beta blockers also reduce IOP by reducing aqueous humor generation. The cardiopulmonary system is known to be impacted by several substances. Timolol and other topical beta blockers have been proven to lower resting blood pressure, spirometry results, and resting pulse rates. There have been reports of cases of severe bradycardia and total cardiac block. There have also been reports of effects on the central nervous system, particularly in elderly people, including disorientation, exhaustion, sadness, and hallucinations. Patients have infrequently mentioned sexual dysfunction as a side effect. The use of topical beta blockers has also been linked to a minimally negative impact on blood lipoproteins. Other infrequently reported adverse effects include concealing thyrotoxicosis signs and concealing hypoglycemia in diabetic individuals.
CARBONIC ANHYDRASE INHIBITORS
Carbonic anhydrase inhibitors (CAIs), like alpha agonists and beta blockers, lower IOP by reducing the formation of aqueous humor. Among other side effects, oral CAIs have been linked to metabolic acidosis, hypokalemia, malaise, anorexia, weight loss, a lack of libido, melancholy, nausea, and digestive distress.
With far fewer systemic adverse effects, topical CAIs have essentially taken the role of oral CAIs in the treatment of glaucoma. Among topical CAIs, brinzolamide and dorzolamide are the two most often used. Topical CAIs frequently produce adverse symptoms including burning at installation, hyperemia, blurred vision, and itching, all of which are likely brought on by the drops’ pronounced acidity (pH 5.5). Some patients report a bitter metallic taste, which may be a result of tear duct drainage into the mouth, where carbonic anhydrase is blocked and leads to an excess of bicarbonate in the saliva.
Because the inhibition of endothelium carbonic anhydrase may result in corneal edema and permanent decompensation in individuals with a history of corneal illness, topical CAIs should be given with caution. After the start of dorzolamide therapy, severe periorbital dermatitis with erythematous, scaly eyelid skin might develop 4 to 40 weeks later.
Bone marrow suppression caused by CAIs, which can result in thrombocytopenia, aplastic anemia, agranulocytosis, pancytopenia, and even mortality, is a seldom described adverse effect. Topical dorzolamide has been known to cause reversible thrombocytopenia. Acetazolamide may also cause ciliochoroidal effusion, which can cause bouts of angle closure and a myopic shift. Last but not least, sulfa-related medications like acetazolamide have been linked to toxic epidermal necrolysis known as Stevens-Johnson syndrome.
PROSTAGLANDIN ANALOGUES
Prostaglandin analogues (PGAs) raise uveoscleral outflow, which lowers IOP. These substances have the potential to cause hypertrichosis, conjunctival redness, irreversible iris darkening, periorbital skin darkening, prostaglandin-associated periorbitopathy, cystoid macular edema, and nongranulomatous anterior uveitis. Typically, hypertrichosis appears 6 to 12 months following the start of PGA. There have also been reports of trichiasis (turned-in eyelashes) needing epilation. Iris and periorbital skin darkening are frequent adverse effects of PGA-induced melanogenesis.
Involutional alterations to the eyelids known as PAP include ptosis (lid drooping), deepening of the lid sulcus, enophthalmos, dermatochalasis, and loss of periorbital fat. Bimatoprost (> 93.3%) may have a very high prevalence of the disease. One month to five years after starting PGA treatment, PAP might appear. The periorbital fat pad has been lost, which is the first discovery.
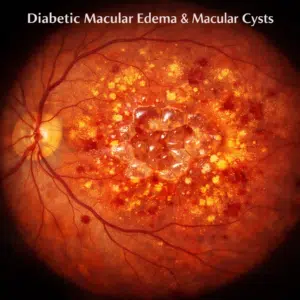
Retrospective analysis revealed that 2.1% of patients receiving latanoprost experienced cystoid macular edema (CME). The problem was linked to present or previous complex or straightforward cataract surgery. With the removal of the PGA or the addition of a topical NSAID, CME can be reversed. 4.1% to 6.4% of eyes may have low-grade nongranulomatous anterior uveitis one to six months after starting latanoprost medication. A topical steroid may be added or the PGA may be stopped in order to treat the uveitis. Patients who are re-challenged with a PGA may have recurrence.
Through the ciliary muscle stiffness caused by prolonged latanoprost usage, which increases uveoscleral outflow, presbyopia may get worse. With the use of PGA, iris cyst development and herpetic keratitis reactivation have also been reported.
The active ingredients latanoprost and nitric oxide are formed when latanoprostene bunod ophthalmic solution (Vyzulta, Bausch + Lomb) is injected into the eye. The trabecular meshwork is relaxed and aqueous outflow is increased by the nitric oxide component. Similar to latanoprost, latanoprostene bunod causes hypertrichosis, conjunctival redness, pigmentation of the iris, and pigmentation of the periorbital skin. Up to this point, rarer problems have not been detailed.
RHO KINASE INHIBITORS
By relaxing smooth muscle, Rho kinase (ROCK) inhibitors promote vasodilation and may also cause conjunctival hyperemia and tiny subconjunctival hemorrhages. One drug in this family, netarsudil ophthalmic solution 0.02% (Rhopressa, Aerie Pharmaceuticals), causes subconjunctival hemorrhages that are often unilateral, tiny, and close to the limbus. In phase 3 studies, 13% of individuals receiving netarsudil experienced subconjunctival hemorrhages. In 9% to 15% of individuals, corneal verticillata developed approximately 2 to 13 weeks after starting netarsudil treatment. Corneal verticillata normally disappears 13 weeks after stopping the medicine and has no effect on visual acuity.
Reticulobullous epithelial edema, commonly referred to as corneal honeycombing, is a less common adverse effect of netarsudil. In a case series, patients had minor anterior chamber inflammation, stromal edema, inferior bullous keratopathy, and decreased visual acuity 2 to 4 weeks after beginning netarsudil treatment. Subcorneal epithelial edema was pronounced and dense. Patients with preexisting risk factors for ocular edema and/or inflammation typically experienced this adverse event. Therefore, before recommending netarsudil, doctors should take these things into account. After stopping the medicine, the edema often went away, but the decrease in visual acuity remained.
Medications and foods to avoid
Medication for glaucoma can help you control the illness and protect your vision. When waiting for surgery, oral medications may be a fantastic interim option. Eye drops might aid with symptom management.
It is important to think about any possible adverse effects of these drugs. Your doctor will inquire about any dietary supplements, herbal treatments, and prescription drugs you are using right now and inform you of any potential interactions.
It is known that grapefruit and a number of widely used drugs can interact. If your doctor has prescribed you medication to treat glaucoma, it is a good idea to consult them about grapefruit eating. However, further research is needed to determine whether or not it causes issues for those taking glaucoma meds.
If you have open-angle glaucoma, you shouldn’t use over-the-counter drugs that make your pupils bigger. Angle-closure glaucoma may occur as a result of them. They consist of the following:
- Antihistamines
- Decongestants
- Motion sickness medication
- Certain antidepressants
Continuing to Use Eye Drops
You must consistently take your glaucoma eye drops as directed by your doctor if you want to prevent irreversible vision loss. During their treatment, some patients have a tendency to repeatedly forget to take their daily ocular pressure drops or, even worse, quit taking their prescriptions completely.
According to a Wilmer Glaucoma Center of Excellence study,
- Each glaucoma patient often barely utilizes 70% of the prescribed eye drops.
- When issued a fresh prescription for glaucoma eye drops, over 25% of patients never get their supply renewed.
- Only 50% of patients who at least once refill their eye drops continue to take their prescribed dosage at the end of the first year.
- Glaucoma eye drops should never be skipped or stopped since this will make the condition worse. Your eye pressure is out of control, causing ongoing irreparable optic nerve damage. The practice can eventually render the afflicted eye permanently blind.
Here are some reminders to help you remember to use your daily dose of glaucoma eye drops as directed:
- Learn to take your medications at the same time as a regular ritual, such as breakfast.
- Track your dosages with a pill-reminder app on your phone, tablet, or wristwatch and receive reminders for your prescription refills on time.
- Ask a family member to remind you if you have any memory problems.
FAQ’s
Which glaucoma drug is least likely to cause adverse effects?
Prostaglandin eye drops are thought to be the most efficient type of glaucoma treatment. Additionally, they only need to be taken once each day, and they often have few adverse effects.
What glaucoma treatment is most popular?
Prostaglandins such as Zioptan, Travatan Z, and Xalatan.
What glaucoma treatment is the safest?
Two of the best drugs now on the market, latanoprostene bunod and netarsudil + latanoprost, have a once-daily dosage and few systemic adverse effects. Both IOPs that are already within the normal-to-low range and IOPs that are significantly higher can be treated with these drugs as a first line.