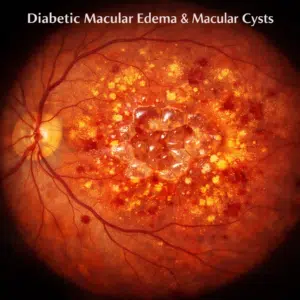
Neovascular age-related macular degeneration (AMD) is one of the main causes of severe vision loss among older people. It occurs when choroidal neovascularization leads to accumulation of fluid and exudates that collect under the macular retina, eventually leading to its degeneration and eventual blindness.
Clinical trials have demonstrated that anti-VEGF medications can greatly enhance functional and anatomic outcomes in those living with exudative AMD, but their use is frequently constrained due to cost concerns.
Efficacy
No matter the advances made in treating AMD, real world patient visual outcomes often fall short of those seen in pivotal phase 3 registration trials. Such trials usually involve injection of drugs that inhibit VEGF; however, frequent visits and monthly injections for such therapy is problematic for many wet AMD patients and thus an extended (T&E) approach is needed as part of therapy to assess its efficacy over a 12-month treatment cycle.
The CATT study is the largest direct head-to-head comparison between two standard intravitreal anti-VEGF agents for treating wet AMD. Participants were randomly divided into one of three treatment arms: PDT with Standard Fluence [SF] + RBZ, DENALI or Sham PDT plus RBZ. After two years, BCVA changes were comparable between these treatment arms. Furthermore, the TREX-AMD trial compared monthly to as-needed administration of RBZ in patients suffering subfoveal CNV secondary to wet AMD.
Safety
Most AMD-related blindness results from its neovascular form, in which abnormal blood vessels form within the macula (central vision). Treatments to inhibit this growth reduces visual loss risk. Many studies have been conducted to study bevacizumab and ranibizumab as potential treatments, including their impact on visual acuity as well as how often they should be given.
These studies included established tests of visual acuity administered by examiners unaware of patients’ treatment assignments, retinal photographs taken before and after treatments were given, and OCT scans assessed at central reading centers. Their results supported using a treat-and-extend approach in treating wet AMD.
However, current intravitreal anti-VEGF-A agent bevacizumab has lower durability compared to earlier agents; new treatments that bind fewer VEGF isoforms may enhance duration of action and durability of these agents. This article details design aspects for next-generation registration trials intended to demonstrate superiority over existing agents.