People suffering from retinitis pigmentosa, a rare genetic eye illness with very few effective treatments, may have reason to be hopeful. Existing medicines only assist a small percentage of the approximate 100,000 Americans who suffer from this disease. However, improvements in gene therapy may soon enable a larger percentage of patients to regain their eyesight.
Retinitis pigmentosa causes the retina’s light-detecting cells to break down over time, resulting in vision loss. This syndrome may be caused by mutations in more than 60 distinct genes. If you’ve been diagnosed with retinitis pigmentosa, you should have genetic testing to find out what’s causing your condition. Because persons with particular genetic variations may be eligible to take part in a clinical trial of a new medicine before it becomes publicly accessible.
Knowing which gene causes your condition and inquiring with your ophthalmologist or optometrist about clinical trial choices might help you save your eyesight.
Causes of Retinitis Pigmentosa
The various kinds of retinitis pigmentosa may be caused by more than 60 distinct genes. There are three methods for parents to pass on the faulty genes to their children:
Autosomal recessive Retinitis Pigmentosa
Each parent has one defective copy and one healthy copy of the relevant gene, but neither shows any symptoms. This kind of retinitis pigmentosa is caused by a kid inheriting two defective copies of the gene (one from each parent). Because the faulty gene requires two copies, each kid in the family has a 1 in 4 chance of being afflicted.
Autosomal dominant Retinitis Pigmentosa
Only one copy of the faulty gene is required for this kind of retinitis pigmentosa to occur. A parent who has that gene has a 50% probability of passing it on to their children.
X-linked RP occurs when a woman possesses the faulty gene and passes it on to her offspring. Each of them has a 50/50 chance of receiving it. The majority of women who possess the gene will not experience any symptoms. However, one out of every five people will have just minor symptoms. The majority of males who get it will have more serious symptoms.
Current Retinitis Pigmentosa Treatments
Retinitis pigmentosa has no cure, but physicians are working hard to identify novel therapies. A few solutions may help you slow down your eyesight loss and even regain a part of your vision:
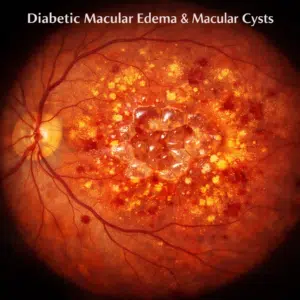
- Swelling of the center of the retina called the macula can occur in the later stages of the disease. This is known as macular edema, and it may also impair your eyesight. Acetazolamide may help you see better and reduce edema.
- Vitamin A: High dosages of this vitamin may halt the progression of retinitis pigmentosa by a year. However, you must exercise caution since too much might be harmful. Follow your doctor’s advice and work closely with him or her.
- Sunglasses: These protect your eyes from dangerous UV radiation that might hasten vision loss by making them less sensitive to light.
- People with late-stage retinitis pigmentosa may be a candidate for a retinal implant, which may restore partial vision. In the United States, the Argus II implant is available. It’s implanted in one eye and combined with a set of camera-equipped spectacles. Electrical pulses are converted from images and transmitted to the retina. Many people were able to find their way to lights and windows. Some patients were able to figure out where other people were in a room, and around half of the participants were able to read lettering up to 9 inches tall.
Newly Approved Retinitis Pigmentosa Treatments
Spark’s vision-restoring gene therapy has been approved by the FDA.
The U.S. Food and Drug Administration has given marketing clearance to Spark Therapeutics’ vision-restoring RPE65 gene therapy, making it the first gene therapy to earn regulatory approval in the United States for the eye or any genetic ailment.
The gene treatment, known as LUXTURNATM (voretigene neparvovec), restored eyesight in persons aged 4 to 44 who had Leber congenital amaurosis (LCA) caused by mutations in the RPE65 gene in a clinical study.
Individuals in the study with severe vision loss said they were able to put their navigational canes aside, see stars, read, and recognize loved ones’ faces. The restoration of vision has lasted more than three years.
Those with retinitis pigmentosa (RP) caused by RPE65 mutations may also benefit from the therapy. The RPE65 gene therapy clinical study at the Children’s Hospital of Philadelphia was made feasible thanks to a $10 million investment by FFB in more than 10 years of lab research (CHOP).
Retinitis Pigmentosa Treatments in Clinical Trials
jCyte Stem-Cell Therapy Enters Phase 3 Clinical Trial for Retinitis Pigmentosa
jCyte, a stem-cell treatment startup, announced positive findings from an 85-person Phase 2b clinical study of its treatment for retinitis pigmentosa patients (RP). In 2021, the business hopes to start a Phase 3 study for the Retinitis pigmentosa treatment. Intravitreal injection of retinal progenitor cells (RPCs), which are stem cells that have partly grown into the retinal cells that allow vision, is used to treat the condition.
Based on lab trials, experts think the therapy may save and perhaps restore eyesight by preserving and potentially rescuing the patient’s existing photoreceptors. The therapy does not involve surgery and may be administered in a matter of minutes in an outpatient setting. The RPCs are injected into the vitreous, which is a gel-like fluid in the eye’s center.
Stem-Cell Therapy from ReNeuron
In a Phase 2 Clinical Trial, the results were encouraging. The clinical study at Massachusetts Eye and Ear Infirmary, ReNeuron, a stem-cell development business based in the United Kingdom, revealed that three patients with retinitis pigmentosa (RP) were treated with stem-cell treatment (MEEI). All of them have seen improvements in their eyesight quickly following therapy. Human retinal progenitor cells (hRPCs) – stem cells that have partly grown into photoreceptors — are injected underneath the patient’s retina as part of the therapy. Researchers expect that the injected hRPCs will integrate into the retina and completely mature into photoreceptors, replacing those lost to illness and therefore restoring vision, based on findings from preclinical experiments. This experiment was made feasible thanks to prior research financed by FFB.
Optogenetic Therapies
By targeting additional retinal cells, optogenetic treatment bypasses standard techniques of healing faulty photoreceptors in the eye. Three recent instances of in-development optogenetic treatments demonstrate many of the obstacles that this sort of gene therapy encounters in the treatment of a terrible genetic eye illness.
By altering cells to produce opsins, which are proteins that transform sensed light into electrochemical signals, optogenetic treatment focuses on retinal cells along the visual pathway. The production of opsin proteins on the surface of these cells, for example, converts them into functional photoreceptor cells, enabling them to detect visible light directly and transmitting those signals to the visual center of the brain.
Clinical studies for GenSight, Bionic Sight, and Nanoscope’s optogenetic therapy for RP and maybe other retinal illnesses have begun. The medicines are intended to help those who are entirely blind due to diseases including retinitis pigmentosa and Usher syndrome. In patients who have lost all of their photoreceptors, the treatment works by giving ganglion cells light sensitivity. In their early-stage testing, Bionic Sight and GenSight both reported some minor vision restoration. For their early-stage study in India, Nanoscope observed visual gains. Nanoscope is now undergoing a Phase 2 study in the United States.
Regardless of their genetic mutation, Gensite’s medication has the ability to benefit patients with retinitis pigmentosa. Optogenetic therapies are treatments that employ light as a tool to manipulate cells.
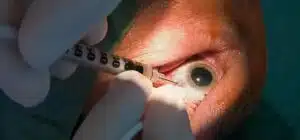
GenSight’s optogenetic treatment involves an eye injection and the use of high-tech eyewear. The injection delivers a gene that aids in the response of retinal cells to light. These cells can transmit electrical messages to the brain thanks to the goggles. The injection and goggles operate together to try to imitate the function of photoreceptors, which are light-sensing cells that don’t perform properly in persons with retinitis pigmentosa.
To learn how to utilize eyewear, patients must practice for many months. Five people have received the therapy thus far. Some people have developed the capacity to recognize high-contrast things on a table and roadway crosswalk lines. They are still unable to read, identify faces, or operate a motor vehicle. Retrosense Therapeutics (Allergan), Nanoscope Therapeutics, and Vedere Bio, Inc. are some of the other businesses working on optogenetic medicines (Novartis).
XLRP Gene Therapy Clinical Trials
Clinical studies for XLRP (RPGR) gene therapy are being conducted by three firms. Biogen is now undergoing a Phase 2/3 experiment. Phase 3 studies are being planned by AGTC and MeiraGTx. In their Phase 1/2 studies, all of the firms showed improvements in eyesight (retinal sensitivity and/or visual acuity). Biogen, on the other hand, failed to satisfy its Phase 2/3 trial’s goals.
Gene Therapy Clinical Trial for RP (PDE6B Mutations)
A Phase 1/2 gene therapy clinical study is being conducted by the French firm Coave for persons with retinitis pigmentosa (RP) caused by PDE6B mutations. A total of 12 patients will be included in the three-year studies at the University Hospital of Nantes in France.
Antioxidant Therapy
Vacuity, located in Dallas, has begun a Phase 1/2 clinical study for its oral antioxidant treatment in Australia. The Foundation Fighting Blindness is funding up to $7.5 million in the development of a potential new treatment for retinitis pigmentosa, Usher syndrome, and other eye diseases. N-acetylcysteine-amide (NACA) is a chemical that protects retinal cells from oxidative stress and hence slows eyesight loss. NACA decreased retinal degeneration in animal models of RP in prior Foundation-funded lab investigations at Johns Hopkins University.
A Sight Saving Protein for a Retinitis Pigmentosa Treatment
SparingVision was founded as a spin-off of the Institut de la Vision with the goal of clinically developing and commercializing a protein known as a rod-derived cone-viability factor (RdCVF). Several prior lab tests financed by the Foundation Fighting Blindness found that the developing medicine was effective. Cones, the cells in the retina that give central and color vision and allow individuals to read, drive, and identify faces, were shown to be protected or delayed by RdCVF. Rods, the retinal cells that give night and peripheral vision, spontaneously release RdCVF. The new retinitis pigmentosa treatment will be the subject of a clinical study in 2021.
Antisense Oligonucleotide for RHO-P23H Mutation
The first patient in ProQR’s Aurora Phase 1/2 clinical trial for autosomal dominant retinitis pigmentosa (adRP) which is caused by the P23H mutation in the gene RHO has received QR-1123, the company’s developing treatment for adRP caused by the P23H mutation in the gene RHO. The variation is the most common cause of adRP in the United States, affecting around 2,500 persons. The research will be conducted at the Retina Foundation of the Southwest in Dallas, Texas, and VitreoRetinal Associates in Gainesville, Florida, with a total of 35 participants.
Conclusion
Effective retinitis pigmentosa treatments have been few and far between in the past but now with the new gene therapies and the mapping of the genome to know where faulty gene mutations are taking place, we have a much brighter future for those suffering from this terrible sight stealing disease. Regular visits to your eye doctor are a must to stay on top of the current technology available to you and get information about enrolling in the ongoing clinical trials. A low vision specialist can help you deal with your current sight loss and maximize your existing vision while participating in the studies.