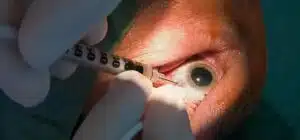
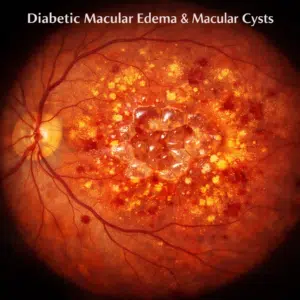
Imagine receiving an eyedrop as opposed to injection directly into your eye to treat age-related macular degeneration (AMD).
It could be conceivable, according to research that was released in the first phase of a trial in 2018. A novel kind of eyedrop that can transport wet AMD therapy to the back of the eye, where the disease destroys the retina and results in blindness, has been created by researchers at the University of Birmingham.
This discovery may significantly alter how AMD, the primary cause of blindness globally, is treated.
“This is intriguing for both patients and practitioners,” said Dr. Jayanth Sridhar, assistant professor of clinical ophthalmology at the Bascom Palmer Eye Institute. “However, this was a preliminary study in animal eyes only. Further research must be conducted on human subjects first to demonstrate safety, and then to establish effectiveness. Still, this study gives at least a glimmer of hope that in the next years we may see a topical drop alternative emerge to supplement or replace the use of intravitreal injections
The need for eye drops for wet macular degeneration
The leading contributor to blindness in industrialized nations is neovascular age-related macular degeneration (nAMD), which exhibits aberrant retinal and choroidal angiogenesis. The main risk factors for nAMD are thought to be endocrine system dysfunctions caused by diabetes and old age.
Since 2006, intravitreal injections of an anti-vascular endothelial growth factor (anti-VEGF) antibody have been the recommended therapy for nAMD. However, the treatment is ineffective and patient compliance is low due to the patient’s anxiety of IVT, long-term periodic injection, and risk of severe IVT events such as hemorrhage, endophthalmitis, retinal detachment, and high intraocular pressure. Consequently, nAMD patients and medical professionals have wished for noninvasive therapy.
Topical eye drops for wet AMD
Due to its non-invasive, self-administering, and user-friendly qualities, topical eye drops for macular degeneration are superior to intravitreal injection. However, inadequate medication exposure at target locations has prevented the use of eye drop products to treat nAMD (retina and choroid). For this reason, numerous recent eye drop pipeline discoveries have been abandoned during the clinical testing phase.
Effective drug delivery capable of overcoming drug removal via ocular clearance, including reflux tears and aqueous humor outflow, and numerous ocular tissue barriers are prerequisites for eye drop administrations for nAMD therapy (such as tightly packed corneal epithelial cells, the blood-aqueous barrier, and blood-retinal barrier).
Due to their ability to block angiogenesis, a number of VEGF tyrosine kinase inhibitors (TKIs) have been authorized for the treatment of cancer. One of these is axitinib, which the US Food and Drug Administration (FDA) authorized in 2012 for the treatment of renal cell carcinoma. This medication is intended to operate as a very effective angiogenesis inhibitor by specifically inhibiting platelet-derived growth factor receptor and vascular endothelial growth factor receptor (VEGFR1, VEGFR2, and VEGFR3) (PDGFR). Axitinib and other VEGF TKIs have been effective in treating cancer, but their limited bioavailability in the posterior eye continues to limit its use in treating nAMD.
Anti-VEGF medicines are now being used to treat wet AMD. It is administered to the eye by a tiny needle by an ophthalmologist, a doctor who focuses on providing both medical and surgical eye treatment. The majority of people may maintain their vision with regular therapy.
The issue is that patients regularly require injections, up to once a month, which makes therapy time-consuming and expensive for both patients and the healthcare system. Complications from the treatment, such as infection and retinal detachment are also possible. Both patients and doctors would prefer a less intrusive option.
Typical eye drops for macular degeneration simply address the surface of the eye. Within minutes the medicine is delivered across the many tissue layers between the surface and the rear of the eye using this novel eyedrop’s cell-penetrating peptide.
A cell-penetrating peptide used in the eye drops technology can carry the medication to the retina (the back of the eye). Anti-VEGF eyedrops administered twice daily to mice were shown by researchers to be just as effective as a single anti-VEGF injection.
Birmingham researchers were collaborating with Macregen Inc. to create a revolutionary line of medicines for AMD and other eye illnesses. Macregen Inc. now owns the scientists’ pending patents for eye drops for macular degeneration.
The joint team was wrapping up the research and was hoping to have clinical trials started by the spring 2019. As of writing this, I have not been able to find any results from those trials.
More recent studies
A study was done by Chinese researchers:
Purpose: Because of the peculiar structure and physiology of the eye, drug distribution to posterior ocular tissues through topical eye drop administration is challenging. Therefore, intravitreal injection or systemic route must be used to provide medicines for posterior eye illness, both of which have disadvantages. This study aimed to show that a carefully formulated eye drop formulation could successfully transport a small-molecule VEGF (vascular endothelial growth factor) inhibitor to the posterior ocular tissues for antiangiogenic treatment.
Methods
A VEGF tyrosine kinase inhibitor, a hydrophilic polymer called hypromellose, and a complex stabilizer called caffeine was used to self-assemble the novel eye drop formulation, known as ITRI AXN eye drops. Non-human Primates (NHPs) and New Zealand White (NZW) rabbits were used in in vivo ocular pharmacokinetics research (NHP). Both pigmented Dutch-Belted rabbits with VEGF-induced retinal neovascularization and Long-Evans rats with laser-induced choroidal neovascularization were used to test the anti-angiogenesis effect.
Results
A drug biodistribution pattern from pharmacokinetic tests suggested that the medication was successfully transported from the ocular surface to the posterior ocular cavity. In the NHP research, excellent drug exposure was shown in the choroid and retina with concentrations 900- and 750-fold higher than the drug IC50 0.5 hours after the eye drop administration (drug level: 0.8%). On illness models in rats and rabbits, the developed formulation likewise showed equivalent antiangiogenic results to intravitreal anti-VEGF antibody injection.
Conclusions
In animal models, our formulation for eye drops has shown tremendous potential as an antiangiogenic treatment against choroidal and retinal neovascularization. The findings indicate that the innovative eye drop formulation can successfully accomplish the goal of this study.
Phase I of SYL1801
Abstract
The goal of this research is to assess the pharmacokinetic (PK) profile, safety, and acceptability of various dosages of SYL1801 ophthalmic solution in healthy volunteers. A crucial regulator of choroidal neovascularization, NRARP (Notch-Regulated Ankyrin Repeat Protein) production is inhibited by SYL1801, a small interfering ribonucleic acid (siRNA) compound.
Methods
The single ascending dose (SAD) and multiple ascending dose (MAD) treatment intervals were used in this observer-masked, parallel groups, time-lagged investigation (MAD). SYL1801 was examined at four distinct dosage levels: 10 mg/mL q.d. 50 mg/mL once day, 25 mg/mL twice daily. both b.i.d (twice daily).
Three participants in each cohort received one therapy day during SAD intervals, whereas six subjects received seven consecutive days of treatment during MAD intervals. One randomly selected eye received eye drop administration on Day 1 (through Day 7 for the MAD interval). Following the final administration, follow-up visits were made 24 and 72 hours later.
Ocular tolerability at the administration sites (cornea and conjunctiva) 72 hours after the last SYL1801 instillation and the assessment of key PK parameters were the primary endpoints. Ocular tolerance at 1 and 24 hours, electrocardiography, laboratory data, secondary PK parameters, and the incidence of adverse events (AE) were also considered secondary outcomes. After the previous injection, PK samples were taken 2, 15, 30 minutes, 1, 4, and 24 hours later (LLOQ: 1 ng/mL).
Results
A total of 36 healthy participants participated in the trial and were assigned to one of the several administration regimens. Four potentially associated adverse events (AEs)—blepharitis in two participants, queratitis, hyperemia, and ocular irritation in one subject each—were recorded in preliminary reports following research close-out; all of them were minor and went away in less than 72 hours. SYL1801 was only found in one research sample, making it unable to evaluate the PK parameters. There was no accumulative impact found.
Conclusions
This study demonstrates the safety and tolerability of SYL1801, a novel siRNA used in eye drops to treat retinal disorders. A good safety profile is supported by low systemic bioavailability and few associated AEs occurrences. This further supports SYL1801’s clinical development, along with data on its effectiveness in both in vitro and in vivo settings.
The 2022 ARVO Annual Meeting, which took place online and in Denver, CO, May 1-4, 2022, both featured this abstract.
Conclusion
The exciting prospect of having an eye drop to treat macular degeneration is on the horizon. It’s just a matter of time before scientists will have a new treatment for this site-threatening disease. The idea that patients will be able to self-treat at home with a drop instead of having monthly injections into their eyes is very exciting to those that have to deal with this unfortunate ailment.
Although nothing is currently available, researchers have made great strides with different methods for this type of drug delivery. Don’t give up hope, stay patient and diligent with your current treatment regimens. It won’t be long before something better is available.
FAQ’s
What are the best eye drops for macular degeneration?
Unfortunately as mentioned above, there currently isn’t an eye drop to treat AMD. You can use artificial tears to help keep the ocular surface in good shape to optimize your vision. If your eyes are dry it will only make your vision worse.
Can macular degeneration be treated with eye drops?
As the studies so far have shown, it is possible to treat macular degeneration with an eye drop. However, the efficacy and delivery mechanisms are not sufficient yet to make this the standard of care. Clinical trials are underway!
What is the newest treatment for macular degeneration?
Another exciting treatment for macular degeneration uses functional medicine, genetic, hormonal, and food allergy testing. More to come on this very effective treatment regime in future blog posts!
How do you prevent macular degeneration from getting worse?
There are many things you can do to prevent AMD from getting worse, and quitting smoking is at the top of the list. Eating a healthy diet of green leafy vegetables, wearing good sun protection, and taking an AREDS formula ocular vitamin are among the best things you can do.
How do you slow down dry macular degeneration?
Aside from the things mentioned in the question above which is primarily for dry macular degeneration, the standard of care currently for wet macular degeneration is an injection of anti-VEGF such as Avastin. This slows the progression of the new blood vessel growth known as neovascularization.
Which drugs are effective against wet macular degeneration?
The most popular medications for treating acute wet macular degeneration now are Avastin, Eylea, Lucentis, and Macugen because they help stop the development of leaky blood vessels in your eye. Although some people may only require therapy once every three months, Lucentis is administered once per month. Every six weeks, Macugen is administered. Following three monthly injections, Eylea is administered once every two months. Although Avastin isn’t precisely an eye medicine, it is frequently used off-label to treat wet macular degeneration.
How do vitamins treat macular degeneration?
Vitamins called antioxidants provide an electron to molecules within our cells that are positively charged. Without this process, oxidation occurs which causes cellular damage and accumulation of metabolic waste. This metabolic waste is called drusen in the retina. Drusen can cause disruptions in the layers of the retina and lead to fluid in the layers of the nerves and photoreceptors causing scarring.
Other vitamins such as Omega 3’s help with cell wall integrity. This keeps our cells healthy and helps to facilitate the process of bringing nutrients such as antioxidants into the cell.